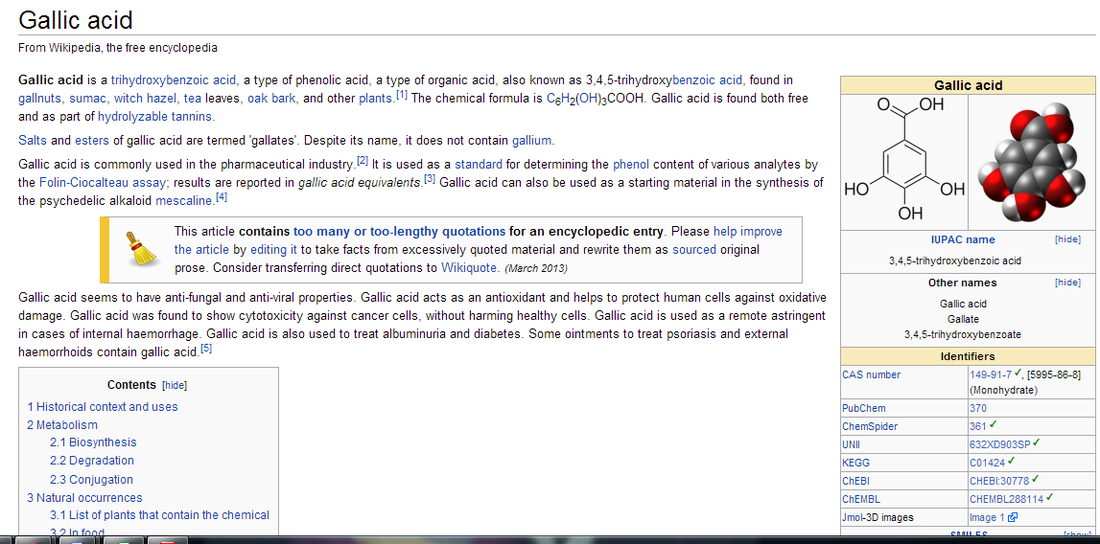
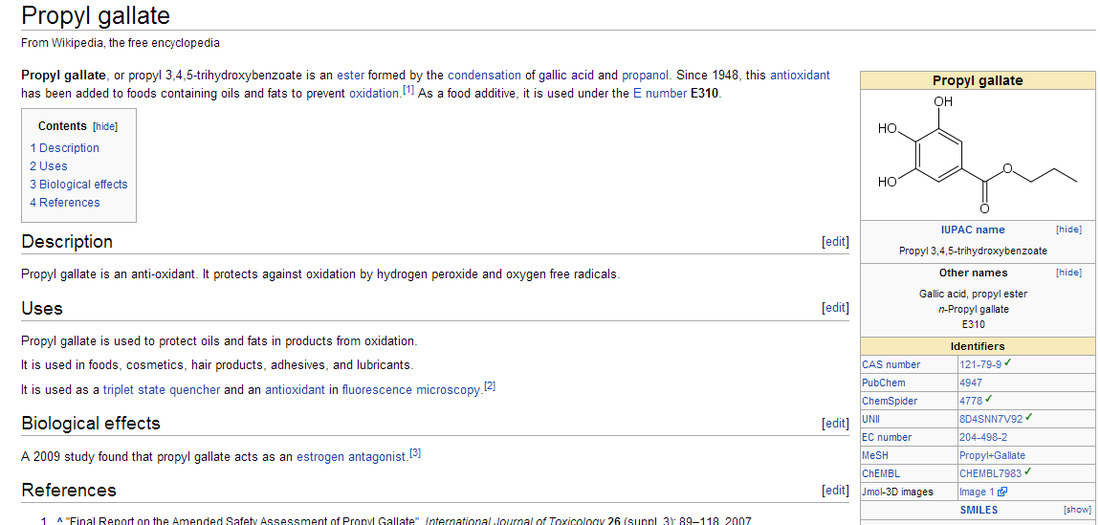
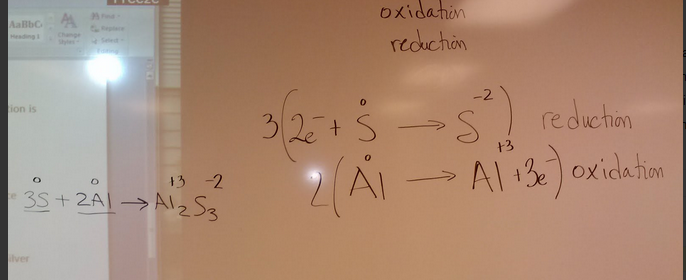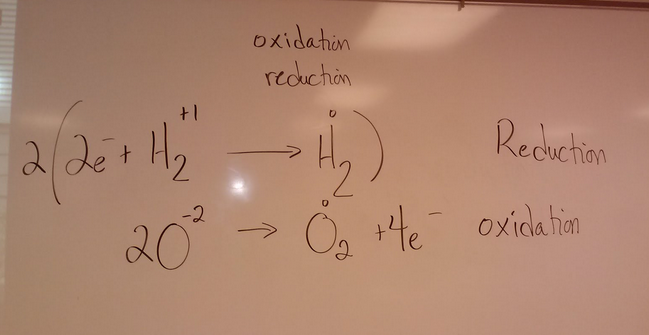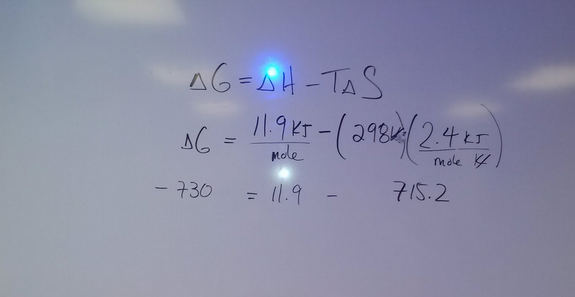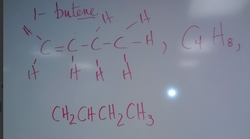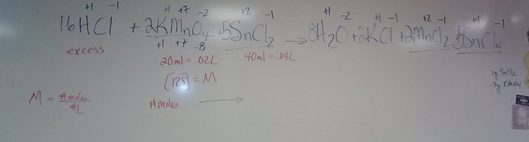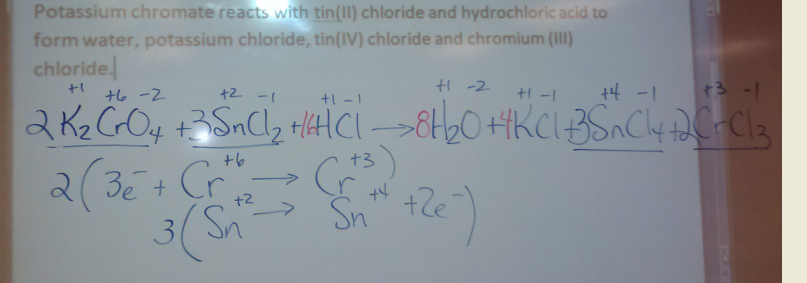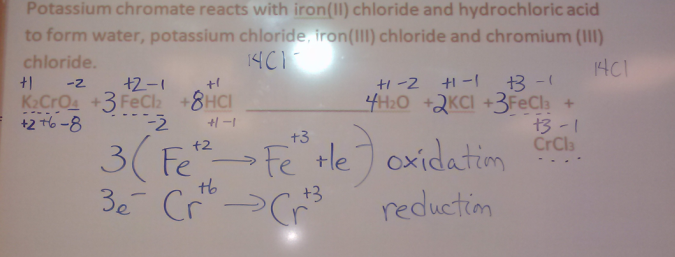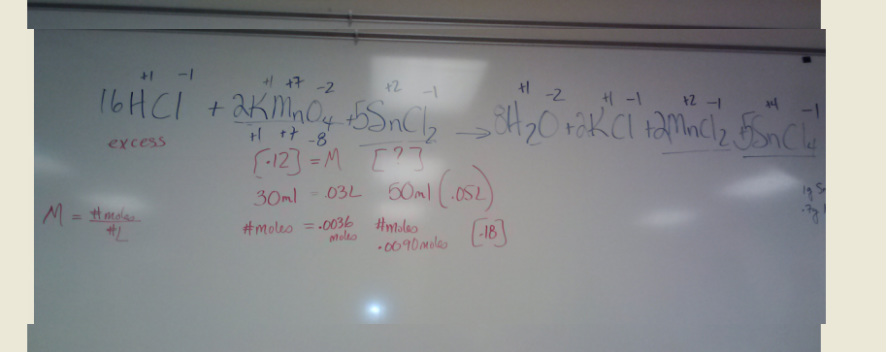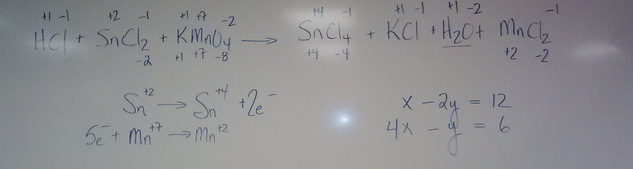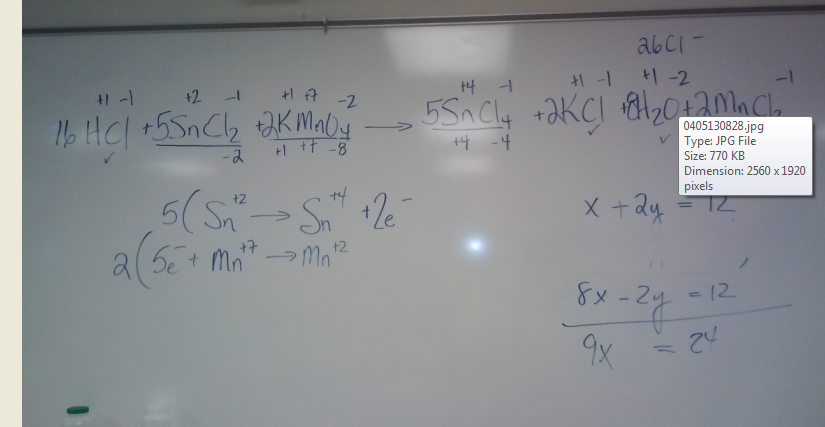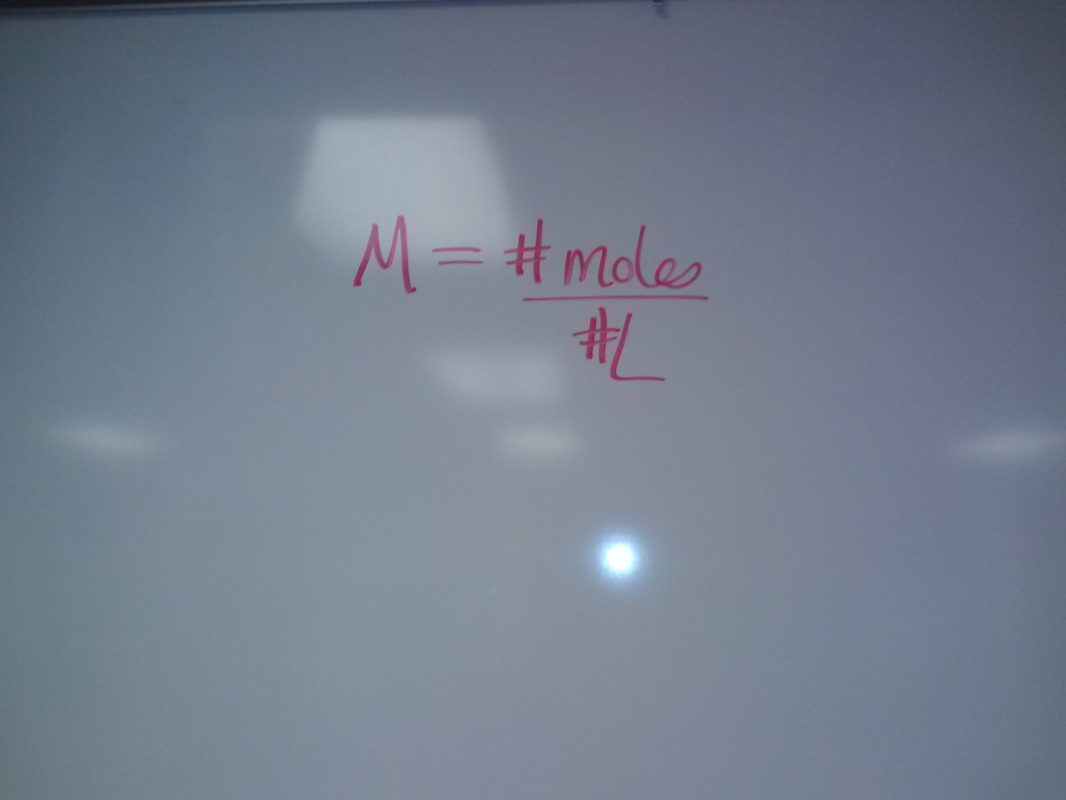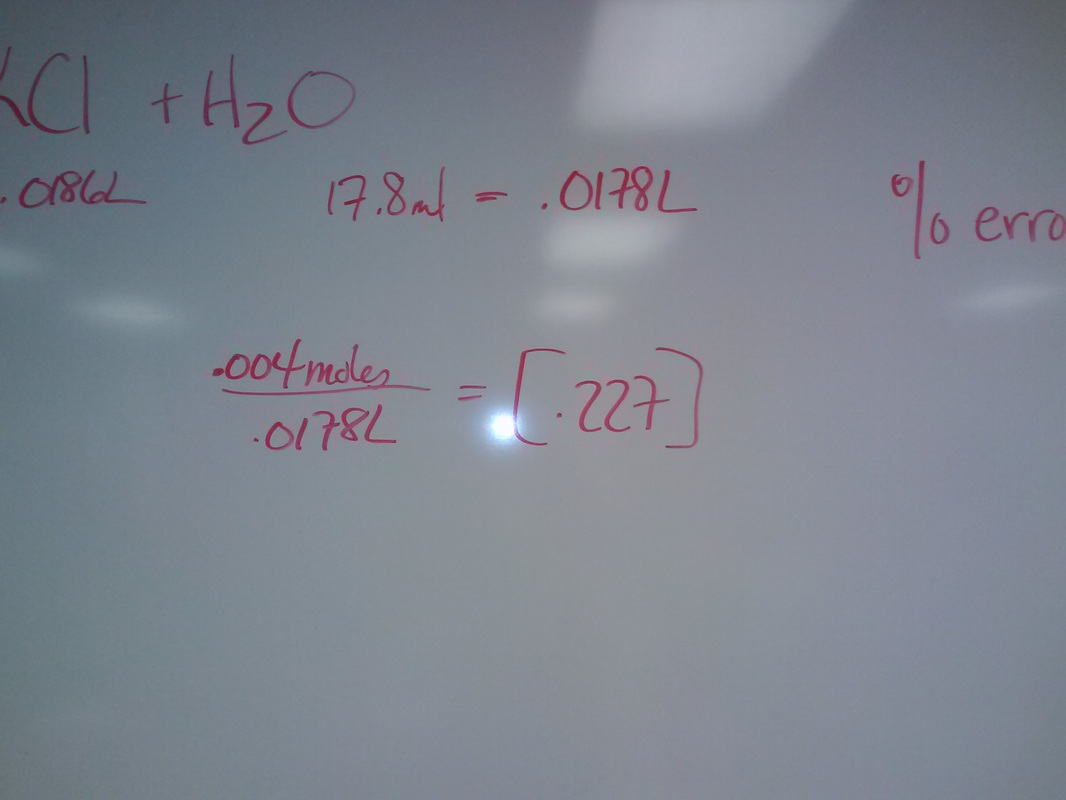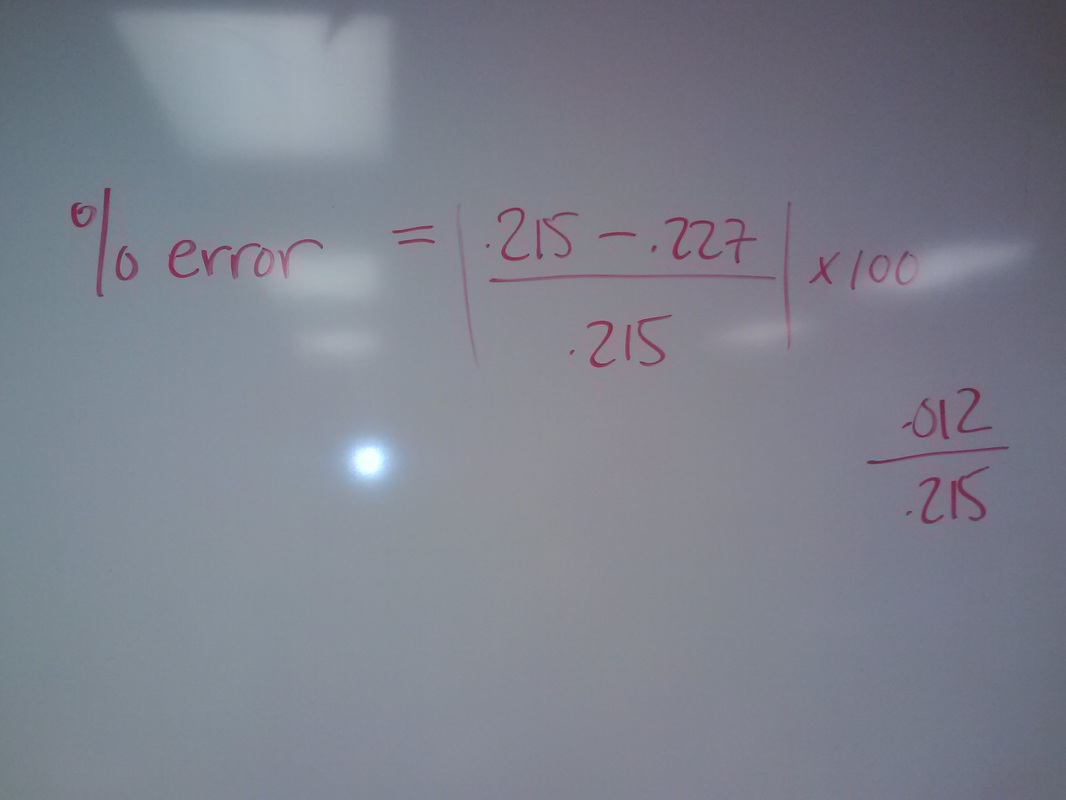Meet Mr. Waterson

Mr. Waterson is a eccentric outgoing teacher who believes in hands on learning. Mr. Waterson is a father of 5 and a grandfather. He taught in Buckley for 19 years before continuing his career in Benzie. He has returned to Buckley as our Chemistry and Physics teacher.
Exam ViewWrite a balanced chemical equation for the following reaction. 5.8 grams of calcium sulfate reacts with excess potassium hydroxide. Figure the following.
Ca(OH)2 + KOH _____________ A. # moles of calcium sulfate reacted B. # moles of potassium hydroxide reacted C. # of grams of each product formed 15.6 grams of propane is burned in 42.8 grams of oxygen to form carbon dioxide and water. Give the following pieces of information. A. balanced chemical equation B. # moles of propane and # moles of oxygen C. Identify the limiting reactant and the excess reactant D. Give the # of grams of each product formed 3. Find the molarity and % by mass of each solution formed A. 21.6 grams of sodium carbonate is dissolved into 140 grams of water to form 155 ml of total solution B. 84.6 grams of lithium phosphate is dissolved in 180 grams of water. The density of the solution is 1.28 g/ml |
Exam ReviewTell if each compound is soluble or insoluble in water
1. sodium phosphate 2. calcium sulfide 3. iron(III) nitrate 4. ammonium carbonate 5. silver chloride 6. nickel(II) oxide 7. aluminum sulfate 8. magnesium chlorate For the following reactions, write out the balanced NET ionic equation. Then, list the spectator ions. A. solutions of sodium carbonate and calcium nitrate are mixed B. solutions of ammonium phosphate and silver chlorate are mixed Draw correct Lewis structures for each of the following molecules. A. ethane B. HCN C. SiO2 D. CH2S E. NCl3 |
Final Exam Review Tuesday 28th
Binary I Binary II Binary III
Ca(NO3)2 Fe2(SO4)3 N2O5
(NH4)2S Al(OH)3
Ca(NO3)2 Fe2(SO4)3 N2O5
(NH4)2S Al(OH)3
| chemistry_review_list_for_exam.docx | |
| File Size: | 11 kb |
| File Type: | docx |
Lab Thursday May 23rd
Chemistry Lab #44 “Batteries”
Purpose: You are to make 4 batteries using different metals.
Procedure: Use a lemon for a source of electrons. Put 2 different metals into the lemon and connect a multimeter to the metals to determine the following.
1. voltage
2. anode
3. cathode
4. positive terminal
5. negative terminal
6. direction of electron flow
Write out the oxidation half reaction and the reduction half reaction for each. Make sure the equation is balanced
Draw and label 1-6 above on a diagram for each setup.
Purpose: You are to make 4 batteries using different metals.
Procedure: Use a lemon for a source of electrons. Put 2 different metals into the lemon and connect a multimeter to the metals to determine the following.
1. voltage
2. anode
3. cathode
4. positive terminal
5. negative terminal
6. direction of electron flow
Write out the oxidation half reaction and the reduction half reaction for each. Make sure the equation is balanced
Draw and label 1-6 above on a diagram for each setup.
Wednesday May 22nd
1. Iron metal reacts with copper (II) sulfate to produce copper metal and iron(II) sulfate. Figure the following
A. oxidation half reaction Fe _______ Fe+2 + 2e-
B. reduction half reaction Cu+2 + 2e- _________Cu
2. Iron metal reacts with copper(II) sulfate to produce copper metal and iron(III) sulfate
A. oxidation half reaction 2 ( Fe ________Fe+3 + 3e-)
B. reduction half reaction 3(Cu+2 + 2e- ___________ Cu)
3. Silver metal reacts with nickel(II) nitrate to produce nickel metal and silver nitrate.
A. oxidation half reaction 2(Ag + 1e- ____________ Ag+)
B. reduction half reaction Ni+2 + 2e- ___________ Ni
A. oxidation half reaction Fe _______ Fe+2 + 2e-
B. reduction half reaction Cu+2 + 2e- _________Cu
2. Iron metal reacts with copper(II) sulfate to produce copper metal and iron(III) sulfate
A. oxidation half reaction 2 ( Fe ________Fe+3 + 3e-)
B. reduction half reaction 3(Cu+2 + 2e- ___________ Cu)
3. Silver metal reacts with nickel(II) nitrate to produce nickel metal and silver nitrate.
A. oxidation half reaction 2(Ag + 1e- ____________ Ag+)
B. reduction half reaction Ni+2 + 2e- ___________ Ni
Tuesday May 21stWrite out equations for each reaction. Tell which elements are changing charges by writing half reactions. Tell which half reaction is oxidizing and which is reducing.
1. Solid elemental sulfur reacts with aluminum metal to produce aluminum sulfide 2. Methane gas burns 3. Copper metal is immersed into a solution of silver nitrate. Silver metal and copper(II) nitrate are produced 4. Electricity is run through water to produce oxygen gas and hydrogen gas. |
Monday May 20th
Balance the following equations. Assume that each is a reaction.
1. Sodium sulfide reacts with calcium chloride
Na2S + CaCl2 _____________
2. Zinc metal reacts with hydrochloric acid to produce hydrogen gas and zinc chloride
Zn + HCl _________
3. Ammonium nitrate reacts with lithium hydroxide to produce ammonia, water and lithium nitrate
NH4NO3 + LiOH ____________
4. Ethanol burns.
C2H5OH + 3 O2 ___________ 2 CO2 + 3 H2O
1. Sodium sulfide reacts with calcium chloride
Na2S + CaCl2 _____________
2. Zinc metal reacts with hydrochloric acid to produce hydrogen gas and zinc chloride
Zn + HCl _________
3. Ammonium nitrate reacts with lithium hydroxide to produce ammonia, water and lithium nitrate
NH4NO3 + LiOH ____________
4. Ethanol burns.
C2H5OH + 3 O2 ___________ 2 CO2 + 3 H2O
Friday May 17th
Chemistry Worksheet 05/17/13
Electricity from Batteries
Explain how a battery produces an electric current
Describe the difference between how a car battery produces and electric current and how a battery you put into your flashlight produces and electric current
Explain what each term means
A. Voltage
B. Amperage
C. Electric charge
D. Electric power
Electricity from Batteries
Explain how a battery produces an electric current
Describe the difference between how a car battery produces and electric current and how a battery you put into your flashlight produces and electric current
Explain what each term means
A. Voltage
B. Amperage
C. Electric charge
D. Electric power
Lab Continued Thur May 16
Lab Wednesday May 15
Chemistry Lab #42 “Free Energy of Baking Soda” Name___________________
Name(s) of Lab Partners_____________________________________________
Purpose: To measure the available energy in a reaction between baking soda and HCl.
Procedure: Measure out exactly 50ml of HCl solution and put into a small Erlenmeyer flask. Record the temperature of the HCl solution. The specific heat of this solution will be the same as water. Measure out exactly 8.0 grams of baking soda in a plastic cup and put into a balloon. Put the balloon tightly over the lip of the Erlenmeyer flask and put the baking soda into the HCl. Measure the circumference of the balloon in cm. Take the balloon off the flask and record the temperature of the solution. Pour the contents of the flask down the sink. Repeat for a 2nd trial.
Materials List:
Observations:
Data Collected: T1 T2 Circumference of balloon
Conclusions: 1. Write out a balanced chemical equation for this reaction
2. Determine the # of moles of baking soda reacted
3. Determine the # of moles of carbon dioxide collected
4. Give the volume in liters of carbon dioxide collected
5. Calculate the pressure inside the balloon
6. Find the # of joules of energy inside the balloon
7. Find the # of Kj/mol of energy inside the balloon
8. Find the # of joules of energy added or subtracted from the solution
9. Calculate the Kj/mol of energy taken from solution
10. Calculate the Free Energy of this reaction using Gibbs Law. Use 298 K for your Temperature
11. Compare your experimental values of H,S and G to the theoretical values found from data on your I-pad
12. Determine the driving force of this reaction. Is it the entropy or the enthalpy?
Name(s) of Lab Partners_____________________________________________
Purpose: To measure the available energy in a reaction between baking soda and HCl.
Procedure: Measure out exactly 50ml of HCl solution and put into a small Erlenmeyer flask. Record the temperature of the HCl solution. The specific heat of this solution will be the same as water. Measure out exactly 8.0 grams of baking soda in a plastic cup and put into a balloon. Put the balloon tightly over the lip of the Erlenmeyer flask and put the baking soda into the HCl. Measure the circumference of the balloon in cm. Take the balloon off the flask and record the temperature of the solution. Pour the contents of the flask down the sink. Repeat for a 2nd trial.
Materials List:
Observations:
Data Collected: T1 T2 Circumference of balloon
Conclusions: 1. Write out a balanced chemical equation for this reaction
2. Determine the # of moles of baking soda reacted
3. Determine the # of moles of carbon dioxide collected
4. Give the volume in liters of carbon dioxide collected
5. Calculate the pressure inside the balloon
6. Find the # of joules of energy inside the balloon
7. Find the # of Kj/mol of energy inside the balloon
8. Find the # of joules of energy added or subtracted from the solution
9. Calculate the Kj/mol of energy taken from solution
10. Calculate the Free Energy of this reaction using Gibbs Law. Use 298 K for your Temperature
11. Compare your experimental values of H,S and G to the theoretical values found from data on your I-pad
12. Determine the driving force of this reaction. Is it the entropy or the enthalpy?
Tuesday, May 14
Write out the balanced chemical equation for the reaction between potassium carbonate and hydrochloric acid to produce water, carbon dioxide and another product.
K2CO3(s) + 2HCl(aq) ____________ H2O(l) + CO2(g) + 2KCl(s)
-1150 2( -167) -286 -394 2( -437) -70kj/mol
156 2 (57) 70 214 2(83) +180 j/mol . K
Write out the balanced chemical equation for the reaction between sodium hydrogen carbonate and hydrochloric acid to produce water, carbon dioxide and another product.
NaHCO3(s) + HCl (aq) __________ H2O(l) + CO2(g) + NaCl(s) +25kj/mol
Write out the balanced chemical equation for the reaction between hydrogen gas and oxygen gas to produce water.
2H2(g) + O2(g) __________ 2 H2O(l)
Standard enthalpy change of formation (data table) From Wikipedia, the free encyclopedia
Jump to: navigation, search
These tables include heat of formation data gathered from a variety of sources, including the primary and secondary literature, as well as the NIST Chemistry WebBook. Note that the table for Alkanes contains ΔfH o values in kcal/mol (1 kcal/mol = 4.184 kJ/mol), and the table for Miscellaneous Compounds and Elements contains these values in kJ/mol.
Contents
Alkanes Formula Name Chemical Formula ΔfH o (kcal/mol) CAS registry number ΔfH o (kJ/mol) H2 Hydrogen [H][H] 0.0 001333-74-0 0.0 CH4 Methane C -17.9 000074-82-8 -74.9 C2H6 Ethane CC -20.0 000074-84-0 -83.7 C2H2 Acetylene CC +54.2 000074-86-2 226.8 C3H8 n-Propane CCC -25.0 000074-98-6 -104.6 C4H10 n-Butane CCCC -30.0 000106-97-8 -125.5 C5H12 n-Pentane CCCCC -35.1 000109-66-0 -146.9 C6H14 n-Hexane CCCCCC -40.0 000110-54-3 -167.4 C7H16 n-Heptane CCCCCCC -44.9 000142-82-5 -187.9 C8H18 n-Octane CCCCCCCC -49.8 000111-65-9 -208.4 C9H20 n-Nonane CCCCCCCCC -54.8 000111-84-2 -229.3 C10H22 n-Decane CCCCCCCCCC -59.6 000124-18-5 -249.4 C4H10 Isobutane (methylpropane) CC(C)C -32.1 000075-28-5 -134.3 C5H12 Neopentane (dimethylpropane) CC(C)(C)C -40.1 000463-82-1 -167.8 C5H12 Isopentane (methylbutane) CCC(C)C -36.9 000078-78-4 -154.4 C6H14 2,2-Dimethylbutane CCC(C)(C)C -44.5 000075-83-2 -186.2 C6H14 2-Methylpentane (isohexane) CCCC(C)C -41.8 000107-83-5 -174.9 C6H14 3-Methylpentane CCC(C)CC -41.1 000096-14-0 -172.0 C6H14 2,3-Dimethylbutane CC(C)C(C)C -42.5 000079-29-8 -177.8 C7H16 2,2,3-Trimethylbutane CC(C)C(C)(C)C -49.0 000464-06-2 -205.0 C7H16 3-ethylpentane CCC(CC)CC -45.3 000617-78-7 -189.5 C8H18 2,2,3,3-Tetramethylbutane CC(C)(C)C(C)(C)C -53.9 000594-82-1 -225.5 C7H16 2-Methylhexane CCCCC(C)C -46.5 000591-76-4 -194.6 C7H16 3-Methylhexane CCCC(C)CC -45.7 000589-34-4 -191.2 C7H16 2,2-Dimethylpentane CCCC(C)(C)C -49.2 000590-35-2 -205.9 C7H16 3,3-Dimethylpentane CCC(C)(C)CC -48.1 000562-49-2 -201.3 C7H16 2,3-Dimethylpentane CCC(C)C(C)C -47.3 000565-59-3 -197.9 C7H16 2,4-Dimethylpentane CC(C)CC(C)C -48.2 000108-08-7 -201.7 C8H18 2-Methylheptane CCCCCC(C)C -51.5 000592-27-8 -215.5 C8H18 2,2-Dimethylhexane CCCCC(C)(C)C -53.7 000590-73-8 -224.7 C8H18 2,3-Dimethylhexane CCCC(C)C(C)C -55.1 000584-94-1 -230.5 C8H18 2,4-Dimethylhexane CCC(C)CC(C)C -52.4 000589-43-5 -219.2 C8H18 2,5-Dimethylhexane CC(C)CCC(C)C -53.2 000592-13-2 -222.6 C8H18 3,3-Dimethylhexane CCCC(C)(C)CC -52.6 000563-16-6 -220.1 C8H18 3,4-Dimethylhexane CCC(C)C(C)CC -50.9 000583-48-2 -213.0 C8H18 3-Ethyl-2-Methylpentane CCC(CC)C(C)C -50.4 000609-26-7 -210.9 C8H18 3-Ethyl-3-Methylpentane CCC(C)(CC)CC -51.4 001067-08-9 -215.1 C8H18 2,2,3-Trimethylpentane CCC(C)C(C)(C)C -52.6 000564-02-3 -220.1 C8H18 2,2,4-Trimethylpentane (isooctane) CC(C)CC(C)(C)C -53.5 000540-84-1 -223.8 C8H18 2,3,3-Trimethylpentane CCC(C)(C)C(C)C -51.7 000560-21-4 -216.3 C8H18 2,3,4-Trimethylpentane CC(C)C(C)C(C)C -51.9 000565-75-3 -217.1 C9H20 3,3-Diethylpentane CCC(CC)(CC)CC -55.7 001067-20-5 -233.0 C9H20 2,2,3,3-Tetramethylpentane CCC(C)(C)C(C)(C)C -56.7 007154-79-2 -237.2 C9H20 2,2,3,4-Tetramethylpentane CC(C)C(C)C(C)(C)C -56.6 001186-53-4 -236.8 C9H20 2,2,4,4-Tetramethylpentane CC(C)(C)CC(C)(C)C -57.8 001070-87-7 -241.8 C9H20 2,3,3,4-Tetramethylpentane CC(C)C(C)(C)C(C)C -56.4 016747-38-9 -236.0 Miscellaneous Compounds
K2CO3(s) + 2HCl(aq) ____________ H2O(l) + CO2(g) + 2KCl(s)
-1150 2( -167) -286 -394 2( -437) -70kj/mol
156 2 (57) 70 214 2(83) +180 j/mol . K
Write out the balanced chemical equation for the reaction between sodium hydrogen carbonate and hydrochloric acid to produce water, carbon dioxide and another product.
NaHCO3(s) + HCl (aq) __________ H2O(l) + CO2(g) + NaCl(s) +25kj/mol
Write out the balanced chemical equation for the reaction between hydrogen gas and oxygen gas to produce water.
2H2(g) + O2(g) __________ 2 H2O(l)
Standard enthalpy change of formation (data table) From Wikipedia, the free encyclopedia
Jump to: navigation, search
These tables include heat of formation data gathered from a variety of sources, including the primary and secondary literature, as well as the NIST Chemistry WebBook. Note that the table for Alkanes contains ΔfH o values in kcal/mol (1 kcal/mol = 4.184 kJ/mol), and the table for Miscellaneous Compounds and Elements contains these values in kJ/mol.
Contents
Alkanes Formula Name Chemical Formula ΔfH o (kcal/mol) CAS registry number ΔfH o (kJ/mol) H2 Hydrogen [H][H] 0.0 001333-74-0 0.0 CH4 Methane C -17.9 000074-82-8 -74.9 C2H6 Ethane CC -20.0 000074-84-0 -83.7 C2H2 Acetylene CC +54.2 000074-86-2 226.8 C3H8 n-Propane CCC -25.0 000074-98-6 -104.6 C4H10 n-Butane CCCC -30.0 000106-97-8 -125.5 C5H12 n-Pentane CCCCC -35.1 000109-66-0 -146.9 C6H14 n-Hexane CCCCCC -40.0 000110-54-3 -167.4 C7H16 n-Heptane CCCCCCC -44.9 000142-82-5 -187.9 C8H18 n-Octane CCCCCCCC -49.8 000111-65-9 -208.4 C9H20 n-Nonane CCCCCCCCC -54.8 000111-84-2 -229.3 C10H22 n-Decane CCCCCCCCCC -59.6 000124-18-5 -249.4 C4H10 Isobutane (methylpropane) CC(C)C -32.1 000075-28-5 -134.3 C5H12 Neopentane (dimethylpropane) CC(C)(C)C -40.1 000463-82-1 -167.8 C5H12 Isopentane (methylbutane) CCC(C)C -36.9 000078-78-4 -154.4 C6H14 2,2-Dimethylbutane CCC(C)(C)C -44.5 000075-83-2 -186.2 C6H14 2-Methylpentane (isohexane) CCCC(C)C -41.8 000107-83-5 -174.9 C6H14 3-Methylpentane CCC(C)CC -41.1 000096-14-0 -172.0 C6H14 2,3-Dimethylbutane CC(C)C(C)C -42.5 000079-29-8 -177.8 C7H16 2,2,3-Trimethylbutane CC(C)C(C)(C)C -49.0 000464-06-2 -205.0 C7H16 3-ethylpentane CCC(CC)CC -45.3 000617-78-7 -189.5 C8H18 2,2,3,3-Tetramethylbutane CC(C)(C)C(C)(C)C -53.9 000594-82-1 -225.5 C7H16 2-Methylhexane CCCCC(C)C -46.5 000591-76-4 -194.6 C7H16 3-Methylhexane CCCC(C)CC -45.7 000589-34-4 -191.2 C7H16 2,2-Dimethylpentane CCCC(C)(C)C -49.2 000590-35-2 -205.9 C7H16 3,3-Dimethylpentane CCC(C)(C)CC -48.1 000562-49-2 -201.3 C7H16 2,3-Dimethylpentane CCC(C)C(C)C -47.3 000565-59-3 -197.9 C7H16 2,4-Dimethylpentane CC(C)CC(C)C -48.2 000108-08-7 -201.7 C8H18 2-Methylheptane CCCCCC(C)C -51.5 000592-27-8 -215.5 C8H18 2,2-Dimethylhexane CCCCC(C)(C)C -53.7 000590-73-8 -224.7 C8H18 2,3-Dimethylhexane CCCC(C)C(C)C -55.1 000584-94-1 -230.5 C8H18 2,4-Dimethylhexane CCC(C)CC(C)C -52.4 000589-43-5 -219.2 C8H18 2,5-Dimethylhexane CC(C)CCC(C)C -53.2 000592-13-2 -222.6 C8H18 3,3-Dimethylhexane CCCC(C)(C)CC -52.6 000563-16-6 -220.1 C8H18 3,4-Dimethylhexane CCC(C)C(C)CC -50.9 000583-48-2 -213.0 C8H18 3-Ethyl-2-Methylpentane CCC(CC)C(C)C -50.4 000609-26-7 -210.9 C8H18 3-Ethyl-3-Methylpentane CCC(C)(CC)CC -51.4 001067-08-9 -215.1 C8H18 2,2,3-Trimethylpentane CCC(C)C(C)(C)C -52.6 000564-02-3 -220.1 C8H18 2,2,4-Trimethylpentane (isooctane) CC(C)CC(C)(C)C -53.5 000540-84-1 -223.8 C8H18 2,3,3-Trimethylpentane CCC(C)(C)C(C)C -51.7 000560-21-4 -216.3 C8H18 2,3,4-Trimethylpentane CC(C)C(C)C(C)C -51.9 000565-75-3 -217.1 C9H20 3,3-Diethylpentane CCC(CC)(CC)CC -55.7 001067-20-5 -233.0 C9H20 2,2,3,3-Tetramethylpentane CCC(C)(C)C(C)(C)C -56.7 007154-79-2 -237.2 C9H20 2,2,3,4-Tetramethylpentane CC(C)C(C)C(C)(C)C -56.6 001186-53-4 -236.8 C9H20 2,2,4,4-Tetramethylpentane CC(C)(C)CC(C)(C)C -57.8 001070-87-7 -241.8 C9H20 2,3,3,4-Tetramethylpentane CC(C)C(C)(C)C(C)C -56.4 016747-38-9 -236.0 Miscellaneous Compounds
Sodium BicarbonateSodium bicarbonate From Wikipedia, the free encyclopedia
Jump to: navigation, search Sodium bicarbonate IUPAC name[hide] Sodium hydrogen carbonate Other names[hide] Baking soda, bicarbonate of soda, nahcolite, sodium bicarbonate, sodium hydrogencarbonate Identifiers CAS number 144-55-8 PubChem 516892 ChemSpider 8609 UNII 8MDF5V39QO EC number 205-633-8 DrugBank DB01390 KEGG C12603 MeSH Sodium+bicarbonate ChEBI CHEBI:32139 ChEMBL CHEMBL1353 RTECS number VZ0950000 ATC code B05CB04,B05XA02, QG04BQ01 Beilstein Reference 4153970 Jmol-3D images Image 1 SMILES [show] InChI [show] Properties Molecular formula NaHCO3 Molar mass 84.007 g mol−1 Appearance White crystals Odor odorless Density 2.20 g cm−3[1] Melting point 50 °C, 323 K, 122 °F (decomposes) Boiling point 851 °C, 1124 K, 1564 °F Solubility in water 9 g/100 mL 69 g/L (0 °C)[2] 96 g/L (20 °C)[3] 165 g/L (60 °C)[3] 236 g/L (100 °C)[2] Solubility Soluble[vague] in acetone, insoluble in ethanol log P -0.82 Acidity (pKa) 10.329[4] 6.351 (carbonic acid)[4] Refractive index (nD) 1.3344 Pharmacology Routes of administration Intravenous, oral Thermochemistry Std enthalpy of formation ΔfHo298 −948 kJ·mol−1[5] Standard molar entropy So298 102 J·mol−1·K−1[5] Hazards MSDS External MSDS Main hazards Causes serious eye irritation NFPA 704 0 1 0 LD50 4.22 g kg− Related compounds Other anions Sodium carbonate Other cations Ammonium bicarbonate Potassium bicarbonate Related compounds Sodium bisulfate Sodium hydrogen phosphate (verify) (what is: /?) Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references Sodium bicarbonate or sodium hydrogen carbonate is the chemical compound with the formula NaHCO3. Sodium bicarbonate is a white solid that is crystalline but often appears as a fine powder. It has a slightly salty, alkaline taste resembling that of washing soda (sodium carbonate). The natural mineral form is nahcolite. It is a component of the mineral natron and is found dissolved in many mineral springs. Since it has long been known and is widely used, the salt has many related names such as baking soda, bread soda, cooking soda, and bicarbonate of soda. In colloquial usage, its name is shortened to sodium bicarb, bicarb soda, or simply bicarb. The word saleratus, from Latin sal æratus meaning aerated salt, was widely used in the 19th century for both sodium bicarbonate and potassium bicarbonate. The term has now fallen out of common usage. Contents |
Thermodynamic ValuesThermodynamic Values at Standard State (298K)
Data Retrieved From: Kots, Treichal, Weaver Chemistry & Chemical Reactivity (Sixth Edition) COPYRIGHT 2006 Species Name Enthalpy "∆Ho" (kJ/mol) Entropy "So" (J/mol*K) Gibbs energy "∆Go" (kJ/mol) Water H2O (l) liquid water -285.83 69.95-237.15 H2O (g) water vapor -241.83188.84 -228.59 Aluminum Al(s) Aluminum solid0 28.3 0 AlCl3 (s)Aluminum Chloride -705.63 109.29 -630.0 Al2O3 (s) Aluminum Oxide -1675.7 50.92 -1582.3 Barium BaCl2 (s) Barium Chloride -858.6 123.68 -810.4 BaCO3 (s) Barium Carbonate -1213112.1 -1134.41 BaO (s)Barium Oxide -548.172.05 -520.38 BaSO4 (s)Barium Sulfate -1473.2132.2 -1362.2 Beryllium Be (s) Beryllium solid 0 9.5 0 Be(OH)2 (s) Beryllium Hydroxide -902.5 51.9 -815.0 Boron BCl3 (g) Boron Trichloride -402.96290.17 -387.95 Bromine Br (g)Bromine gas 111.884 175.022 82.396 Br2 (l)Bromine liquid 0152.2 0 Br2 (g)Bromine gas 30.91 245.47 3.12 BrF3 (g)Bromine Triflouride -255.60 292.53 -229.43 HBr (g)Hydrogen Bromide gas-36.29 198.70 -53.45 Calcium Ca (s) Calcium 041.59 0 Ca (g) Calcium178.2 158.884 144.3 Ca2+ (g) Calcium (II) ion1925.90 ---- ---- CaC2 (s) Calcium Carbide-59.8 70 -64.93 CaCO3 (s, calcite) Calcium Carbonate-1207.6 91.7 -1129.16 CaCl2 (s) Calcium Chloride-795.8 104.6 -748.1 CaF2 (s)Calcium Flouride -1219.668.87 -1167.3 CaH2 (s) Calcium Hydride-186.2 42 -147.2 CaO (s)Calcium Oxide -635.09 38.2 -603.42 CaS (s)Calcium Sulfide -482.4 56.5 -477.4 Ca(OH)2 (s) Calcium Hydroxide solid-986.09 83.39-898.43 Ca(OH)2 (aq) Calcium Hydroxide aqueous -1002.82 ---- -868.07 CaSO4 (s) Calcium Sulfate-1434.52 106.5 -1322.02 |
Friday May 10th |
|
|
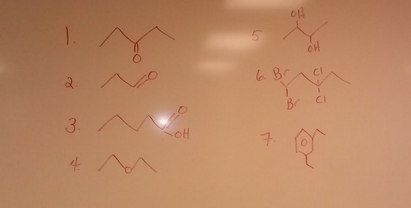
Draw a correct structure for the following organic compounds. Then, give a correct molecular formula for each.
1. Pentanal 2. Butanone 3. Propanoic acid 4. 1,1,1 – tri hydroxyl butane 5. 3- propanone 6. Benzoic acid 7. Ethanal 8. Propyl benzene 9. 2,2 – dibromo – 3,4 – diflurohexane 10. Diethyl ether |
|
Acid's Friday May 3rd
Thursday May 2ndDraw a correct structure for the following organic compounds
1. 1,2,4-trimethyl benzene 2. 1-ethyl-2-methyl benzene 3. 1-butanol 4. 1,2-pentandiol 5. cyclopentene 6. 1,1-dimethyl cyclobutane 7. oxalic acid 8. Butanoic acid 9. Tartaric acid 10. Citric acid |
Websites |
|
Monday April 29, 2013
There is a organic chemical category called aromatics. Please look up what kind of compounds make up aromatics and in particular, look up both the structure and formula for benzene. Then, give some uses for aromatics and some problems with aromatic compounds.
Look up the formula and draw a structure for the following.
1. toluene 2. Nitrobenzene 3. Xylene 4. Cyclohexane 5. cyclobutane
Look up the formula and draw a structure for the following.
1. toluene 2. Nitrobenzene 3. Xylene 4. Cyclohexane 5. cyclobutane
Thursday April 25, 2013
http://www.cosmolearning.com/video-lectures/alkynes/
Carbon
http://www.cosmolearning.com/video-lectures/alkanes/
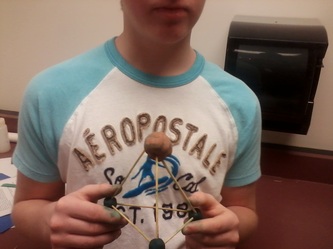
Chemistry Lab for 4/23/13

You will need to build models of 5 molecules using clay to represent your carbon atoms and bamboo sticks to represent bonds. You will not need to represent each hydrogen atom.
Make models of the 5 isomers of C4H8. Take pictures of each model and email the pictures to me. Make sure that each model represents the 3-D model of each. You only need 1 set of pictures for each group.
Make models of the 5 isomers of C4H8. Take pictures of each model and email the pictures to me. Make sure that each model represents the 3-D model of each. You only need 1 set of pictures for each group.
Monday April 22, 2013
Friday April 19th
Carbon bondingGive the formula and draw the structure for each of the following molecules
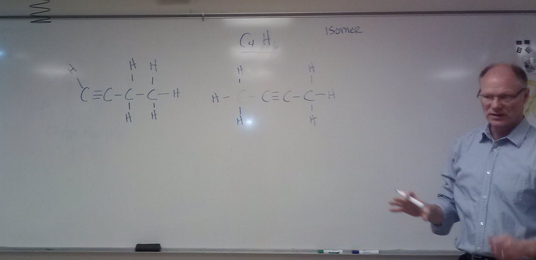
A. Butyne C4H6 B. Pentyne C5H8 C. Hexyne What would the formula for an alkyne with 20 carbons be? Isomer- same formula, different structure Visit NBCNews.com for breaking news, world news, and news about the economy |
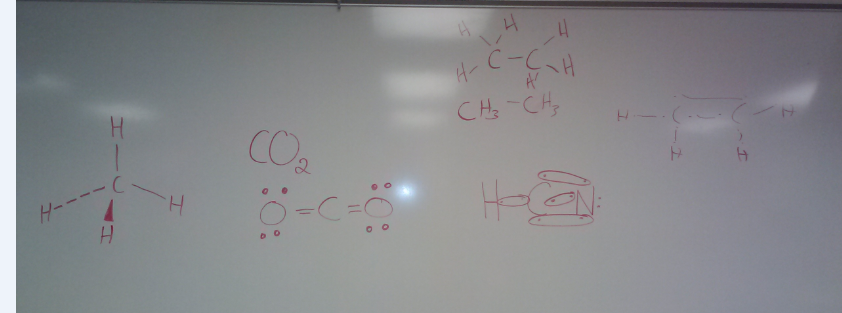
Thursday April 18thDraw out Lewis structures for the following molecules.
A. methane B. carbon dioxide C. ethene D. hydrogen cyanide E. ethene F. ethyne (acetylene) G. G. propane, propene, propyne 4 ways to describe organic molecules. 1. molecular formula 2. condensed formula 3. skeletal structure 4. Lewis structure |
Wednesday April 17th
Explain the difference between an organic molecule and an inorganic molecule.
What 2 elements are common to organic molecules?
Numerical prefixes are different for organic molecules than they are for inorganic molecules. For example, inorganic molecules have prefixes of mono = 1, di = 2 etc. What are the 1st 10 prefixes for organic molecules?
Tell whether each molecule is organic or inorganic.
A. water B. methane C. acetylene D. carbon dioxide E. oxygen
F. alcohol G. starting fluid H hydrogen peroxide
What 2 elements are common to organic molecules?
Numerical prefixes are different for organic molecules than they are for inorganic molecules. For example, inorganic molecules have prefixes of mono = 1, di = 2 etc. What are the 1st 10 prefixes for organic molecules?
Tell whether each molecule is organic or inorganic.
A. water B. methane C. acetylene D. carbon dioxide E. oxygen
F. alcohol G. starting fluid H hydrogen peroxide
Lab
Chemistry Lab # 27 04/16/13
Procedure: Measure out exactly 40 ml of hydrogen peroxide solution into a beaker. Add 20ml of HCl to the same beaker. Put potassium permanganate into your buret and then titrate the solution in the beaker with the potassium permanganate until the endpoint is reached. Discard the solution and repeat this procedure for a 2nd trial. You must wear SAFETY GLASSES.
Procedure: Measure out exactly 40 ml of hydrogen peroxide solution into a beaker. Add 20ml of HCl to the same beaker. Put potassium permanganate into your buret and then titrate the solution in the beaker with the potassium permanganate until the endpoint is reached. Discard the solution and repeat this procedure for a 2nd trial. You must wear SAFETY GLASSES.
Balancing Equations Wednesday April 10Balance the following equation
20ml [.125] potassium permanganate reacts with 40 ml of tin(II) chloride to reach a pink endpoint. You add HCl to the tin(II) chloride before you put in the 20 ml of potassium permanganate. Give the molarity of the tin(II) chloride. |
Balancing Complex Equations
Following the Trail of Electrons
Helpful Website
Tuesday April 2nd
NaCl Na = +1 (oxidation number) Cl = -1 sodium chloride
AlF3 Al = +3 F = -1 aluminum fluoride
FeO Fe = +2 O = -2 iron (II) oxide
CrBr6 Cr = +6 Br = -1 chromium (VI) bromide
Ca3N2 Ca = +2 N = -3 calcium nitride
NBr3 N = +3 Br = -1 or is it N = -3 Br = =+1 nitrogen tribromide
SrF2
Ni2S3
ClF3
Ag3P
NH3
PbO2
AlF3 Al = +3 F = -1 aluminum fluoride
FeO Fe = +2 O = -2 iron (II) oxide
CrBr6 Cr = +6 Br = -1 chromium (VI) bromide
Ca3N2 Ca = +2 N = -3 calcium nitride
NBr3 N = +3 Br = -1 or is it N = -3 Br = =+1 nitrogen tribromide
SrF2
Ni2S3
ClF3
Ag3P
NH3
PbO2
Tuesday March 19, 2013
A solution of calcium chloride reacts with solid magnesium carbonate to form solid magnesium chloride and solid calcium carbonate.
Liquid ethanol burns with oxygen to create carbon dioxide and liquid water.
Solid potassium oxide reacts with a solution of hydrochloric acid to form solid potassium chloride and liquid water.
Liquid ethanol burns with oxygen to create carbon dioxide and liquid water.
Solid potassium oxide reacts with a solution of hydrochloric acid to form solid potassium chloride and liquid water.
Monday March 18, 2013
A solution of calcium chloride reacts with solid magnesium carbonate to form a solution of magnesium chloride and solid calcium carbonate.
CaCl2 + MgCO3 ________________ CaCO3 + MgCl2
Liquid ethanol burns with oxygen to create carbon dioxide and liquid water.
C2H6O + 3 O2 ________________ 2 CO2 + 3 H2O
Solid potassium oxide reacts with a solution of hydrochloric acid to form solid potassium chloride and liquid water.
K2O + 2 HCl _______________ 2 KCl + H2O
CaCl2 + MgCO3 ________________ CaCO3 + MgCl2
Liquid ethanol burns with oxygen to create carbon dioxide and liquid water.
C2H6O + 3 O2 ________________ 2 CO2 + 3 H2O
Solid potassium oxide reacts with a solution of hydrochloric acid to form solid potassium chloride and liquid water.
K2O + 2 HCl _______________ 2 KCl + H2O
Friday March 15, 2013
Starting Fluid is diethyl ether. The formula for diethyl ether is
As you know, starting fluid burns in air. Write out a balanced chemical equation for the burning of starting fluid.
You 3.8 grams of starting fluid. How much heat is released if it releases 2700kj/mole?
C4H10O + O2____________________ CO2 + H2O
A diesel fuel molecule is C15H32. Write out a balanced chemical equation for the burning of diesel fuel.
C15H32 + O2 ____________________ CO2 + H2O
As you know, starting fluid burns in air. Write out a balanced chemical equation for the burning of starting fluid.
You 3.8 grams of starting fluid. How much heat is released if it releases 2700kj/mole?
C4H10O + O2____________________ CO2 + H2O
A diesel fuel molecule is C15H32. Write out a balanced chemical equation for the burning of diesel fuel.
C15H32 + O2 ____________________ CO2 + H2O
Disappearing job market

Entry level jobs are disappearing to machines
Thursday March 14, 2013
Look up the formula for ethanol.
Ethanol burns when oxygen gas is added to form carbon dioxide and water. Write out the chemical equation for this reaction.
Look up the formula for gasoline. (octane) Write out the balanced equation for the burning of gasoline with oxygen to produce carbon dioxide and water.
Which is more efficient?
C2H6O + 3 O2 _______________ 2 CO2 + 3 H2O
1 gallon = 1370 kj/mole
C8H18 + 12.5 O2__________________ 8 CO2 + 9 H2O
1 gallon = 5510 kj/mole
Ethanol burns when oxygen gas is added to form carbon dioxide and water. Write out the chemical equation for this reaction.
Look up the formula for gasoline. (octane) Write out the balanced equation for the burning of gasoline with oxygen to produce carbon dioxide and water.
Which is more efficient?
C2H6O + 3 O2 _______________ 2 CO2 + 3 H2O
1 gallon = 1370 kj/mole
C8H18 + 12.5 O2__________________ 8 CO2 + 9 H2O
1 gallon = 5510 kj/mole
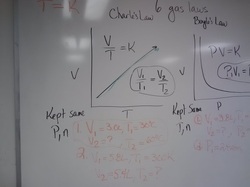
Tuesday March 12, 2013Explain how you could measure the heat lost or gained in a chemical reaction.
Draw a graph with X and Y axes. Graph out an exothermic process and an endothermic process. Show activation energy on these graphs. Write out the equation for calculating how much heat is released in a chemical reaction. Label each quantity and give units. Give a definition for specific heat. Give the specific heat of water. I will hand out a lab sheet to you. Take notes for the lab tomorrow. |
Monday March 11Explain the difference between an exothermic and an endothermic reaction or process.
Explain how you could measure the heat lost or gained in a chemical reaction. Draw a graph with X and Y axes. Graph out an exothermic process and an endothermic process. Show activation energy on these graphs. |
|
Friday March 8Doppler Effect Change in frequency based on the sound movement. 30mi/hr and the car horn had a frequency of 420hz. What frequency did we hear as the car came towards us and what frequency did we hear as the car went away from us?
325m/s speed of sound in air. Car speed was 13.4 m/s Wave speed = wavelength X frequency wavelength = .77m 325 + 13.4 =( .77m) (f) Frequency of the horn wave going away from us. |
|
Acid and Bases
Sample problems
1. NaOH = 16 + 1 + 23 = 40g/mole so…. 1.2g/40g/mole = .03 moles
2. Molarity = # moles/ # liters solution so…… .03 moles/ .500L = [.06]
(I changed 500ml to liters by dividing by a 1000)
3. pH = -log[H+] so….. pH = -log[.06] = 1.22 However, NaOH is a base so…. You subtract the 1.2 from 14. This gives a pH of 12.8
2. Molarity = # moles/ # liters solution so…… .03 moles/ .500L = [.06]
(I changed 500ml to liters by dividing by a 1000)
3. pH = -log[H+] so….. pH = -log[.06] = 1.22 However, NaOH is a base so…. You subtract the 1.2 from 14. This gives a pH of 12.8
Chemistry Quiz 3/1/13
Chemistry Quiz 03/01/13 Name________________________
Name each of the following acids
1. HNO3 2. HCl 3. HBrO3
4. HI 5. H2CO3 6. HClO3
Give correct formulas for each acid name
1. sulfurous acid 2. Hydroselenic acid
3. chromic acid 4. Acetic acid
5. hydrofluoric acid 6. Nitrous acid
You measure out exactly 1.56 grams of hydrochloric acid (HCl) and dissolve into enough water to make 4.0 liters of solution. Give the following
A. # moles of HCl
B. molarity of this solution
C. pH of this solution
You measure out 0.25 grams of sodium hydroxide and dissolve into enough water to make 600ml of solution. Give the following
A. # moles of sodium hydroxide
B. molarity of this solution
C. pH of this solution
2nd hour will have an extra sheet to attach to this quiz
Name each of the following acids
1. HNO3 2. HCl 3. HBrO3
4. HI 5. H2CO3 6. HClO3
Give correct formulas for each acid name
1. sulfurous acid 2. Hydroselenic acid
3. chromic acid 4. Acetic acid
5. hydrofluoric acid 6. Nitrous acid
You measure out exactly 1.56 grams of hydrochloric acid (HCl) and dissolve into enough water to make 4.0 liters of solution. Give the following
A. # moles of HCl
B. molarity of this solution
C. pH of this solution
You measure out 0.25 grams of sodium hydroxide and dissolve into enough water to make 600ml of solution. Give the following
A. # moles of sodium hydroxide
B. molarity of this solution
C. pH of this solution
2nd hour will have an extra sheet to attach to this quiz
Feb 27thFind the logarithm for each number
1. 350 2. 8.6 3. 5 4. 50 5. 500 6. .005 7. 1.8 8. .0000064 Give a number associated with each logarithm. 1. 2.4 (10^2.4) 2. -1.55 3. .36 4. -2.88 5. 3.9 6. -4.02 |
Molarity and PHYou measure out exactly 2.4 grams of HCl and dissolve into enough water to make 5.0 liters of solution. Give the following
A. molarity of this solution B. pH of this solution You measure out exactly .36 grams of potassium hydroxide into enough water to make 2.50 liters of solution. Give the following A. Molarity of this solution B. pH of this solution pH= -log(acid) |
Monday Feb 25, 2013
Distinguish between binary acids and oxyacids
1. HBr
1. HBr
2
Molarity Lab
Molarity 1/29/13
Mr. Kent's Chemistry Page
|
|
|
01/18/13 Chemistry Lab
Find 10 reactions, be sure to use your solubility rules
Solubility Guidlines
- All common compounds of Group I and ammonium ions are soluble.
- All nitrates, acetates, and chlorates are soluble.
- All binary compounds of the halogens (other than F) with metals are soluble, except those of Ag, Hg(I), and Pb. Pb halides are soluble in hot water.)
- All sulfates are soluble, except those of barium, strontium, calcium, lead, silver, and mercury (I). The latter three are slightly soluble.
- Except for rule 1, carbonates, hydroxides, oxides, silicates, and phosphates are insoluble.
- Sulfides are insoluble except for calcium, barium, strontium, magnesium, sodium, potassium, and ammonium.
| chemistry_exam_1st_semester________name.docx | |
| File Size: | 11 kb |
| File Type: | docx |
January 9, 2013
| chemistry_carbon_dioxide_release.docx | |
| File Size: | 10 kb |
| File Type: | docx |
IDEAL GAS EQUATION 1-7-13
Gas Law

Use this law with your CO2 pop lab to find amount of CO2 in pop
Mr. Waterson's Chemistry Class
11/26/12 (Hand this assignment in)
Give the SHORTHAND electron configuration and orbital notation for each of the following atoms
1. N
2. S
3. Ca
4. Ar
5. Si
6. K
Give the LEWIS Diagram for each element
11/19/12
Give the following quantities for Li, Mg and Cl
1. #p
2. #n
3. #e
4. Electron analysis
Li 3p, 4n, 3e 2-1
Mg 12p, 12n,12e 2-8-2
Cl 17p, 18n, 17e 2-8-7
Look up the following definitions and make sure you know how to apply them. Na, F, Ca
1. Electron configuration for electrons
Na 1s²2s²2p⁶3s²
F 1s²2s²2p⁵
Ca 1s²2s²2p⁶3s²3p⁶4s²
2. Orbital notation for electrons
3. Lewis diagrams for electrons
10/30/12 Home Work React 8.6 grams of Magnesium Chloride with Lithium Sulfate
Write an balanced equation
Determine Molar Mass
How many grams of lithium sulfate would be needed?
Give the SHORTHAND electron configuration and orbital notation for each of the following atoms
1. N
2. S
3. Ca
4. Ar
5. Si
6. K
Give the LEWIS Diagram for each element
11/19/12
Give the following quantities for Li, Mg and Cl
1. #p
2. #n
3. #e
4. Electron analysis
Li 3p, 4n, 3e 2-1
Mg 12p, 12n,12e 2-8-2
Cl 17p, 18n, 17e 2-8-7
Look up the following definitions and make sure you know how to apply them. Na, F, Ca
1. Electron configuration for electrons
Na 1s²2s²2p⁶3s²
F 1s²2s²2p⁵
Ca 1s²2s²2p⁶3s²3p⁶4s²
2. Orbital notation for electrons
3. Lewis diagrams for electrons
10/30/12 Home Work React 8.6 grams of Magnesium Chloride with Lithium Sulfate
Write an balanced equation
Determine Molar Mass
How many grams of lithium sulfate would be needed?
Rutherford's 4 Conclusions

WHAT YOU NEED TO KNOW

You need to be able to do:
- Write a Balanced Equation
- Determine Molar Mass
- Give percent of each element
- Give Grams of each element
- Determine Moles
Balancing Chemical Equations |
|